Therapeutic peptides and exosomes are both being deployed in regenerative medicine clinics with increasing frequency. Both are biologically active, both produce measurable clinical effects, and both are being presented to patients as tools for cellular rejuvenation and anti-aging. But they are not the same class of intervention — and understanding the distinction is the difference between a targeted pharmacological signal and a regenerative biological cascade.
One tells a cell what to do. The other gives the cell the tools to rebuild itself. This article examines the science behind each, compares their clinical applications and limitations, and explains how XELGEN's DNA methylation platform provides the objective measurement layer that allows clinicians to determine which intervention is actually moving the biological age needle — and by how much.
What Are Therapeutic Peptides?
Peptides are short chains of amino acids — typically 2 to 50 residues in length — that act as signalling molecules in the body. Endogenous peptides regulate virtually every physiological process, from hormone release and immune function to tissue repair and appetite. Therapeutic peptides are either synthetic analogues of naturally occurring peptides or novel sequences designed to mimic or modulate specific biological pathways.
| Peptide | Primary Mechanism | Clinical Application |
|---|---|---|
| BPC-157 | Promotes angiogenesis, activates growth hormone receptors, modulates nitric oxide | Tissue repair, gut healing, tendon and ligament regeneration |
| TB-500 (Thymosin β4) | Upregulates actin polymerisation, promotes cell migration and differentiation | Wound healing, muscle repair, anti-inflammatory |
| GHK-Cu (Copper peptide) | Activates wound healing genes, promotes collagen synthesis, antioxidant | Skin rejuvenation, hair growth, tissue remodelling |
| CJC-1295 / Ipamorelin | GHRH analogue; stimulates growth hormone pulse | Muscle mass, fat metabolism, sleep quality, recovery |
| Epithalon | Telomerase activator, regulates pineal gland function | Telomere maintenance, circadian rhythm, anti-aging |
How Peptides Work: Targeted Receptor Binding
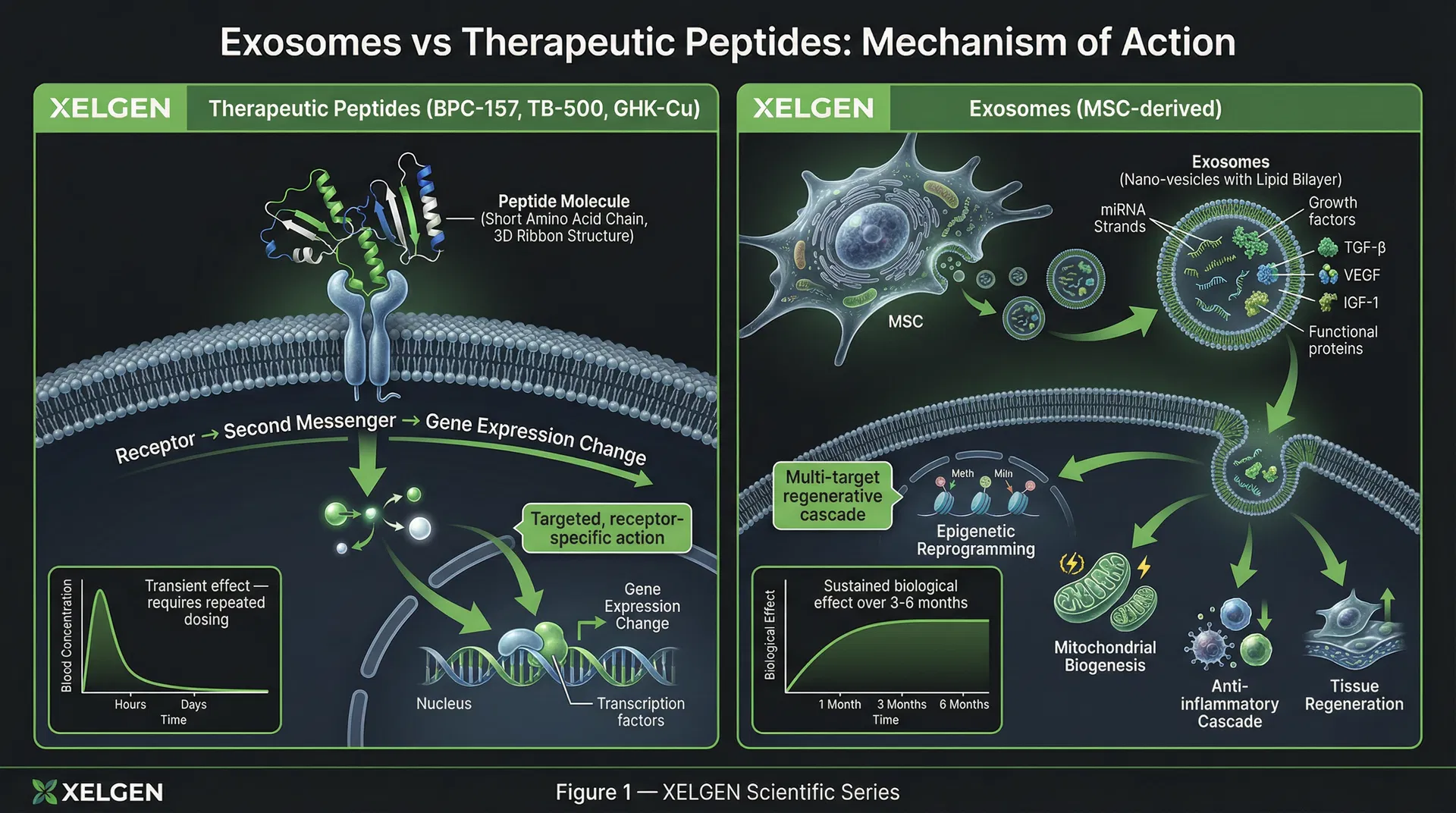
The defining characteristic of therapeutic peptides is specificity. Each peptide binds to a defined receptor or set of receptors on the cell surface, triggering a downstream intracellular signalling cascade. This cascade typically involves second messengers (cAMP, IP3, DAG), kinase activation, and ultimately changes in gene expression via transcription factor modulation.
This specificity is both the greatest strength and the fundamental limitation of peptide therapy. A peptide designed to stimulate growth hormone release will do exactly that — and nothing else. BPC-157 will promote angiogenesis and activate healing pathways in the tissue where it is administered. These are precise, reproducible, pharmacologically predictable effects. But they are also transient. Once the peptide is cleared from circulation — typically within hours to days — the signalling stimulus ends. The downstream gene expression changes begin to revert. The biological effect fades.
⚠ The Fundamental Constraint
Peptides are tools, not teachers. They instruct the cell to perform a specific function for as long as the signal persists. They do not reprogram the cell's fundamental operating state. Without continued dosing, the cell returns to its pre-intervention baseline.
What Are Exosomes?
Exosomes are extracellular vesicles ranging from 30 to 150 nanometres in diameter, secreted by virtually all cell types as part of normal intercellular communication. They are not simply fragments of cellular debris — they are deliberately packaged biological messengers, loaded with a specific cargo that reflects the functional state of the cell that produced them.
The cargo of a therapeutic exosome preparation — particularly those derived from mesenchymal stem cells (MSCs) — typically includes microRNAs (miRNAs) that regulate entire gene networks, messenger RNAs that can be translated in recipient cells, growth factors including TGF-β, VEGF, IGF-1, and HGF, functional proteins such as enzymes and transcription factors, and bioactive lipid mediators. When an exosome fuses with the membrane of a target cell and delivers this cargo, the effect is categorically different from peptide receptor binding. The cell is not receiving a single instruction through a single receptor. It is receiving a comprehensive biological programme.
The Paracrine Cascade: Amplification Beyond the Initial Target
One of the most clinically significant properties of exosome therapy is the paracrine cascade it initiates. When MSC-derived exosomes are administered intravenously, the regenerative signals they deliver propagate outward — treated cells begin producing their own exosomes and signalling molecules, which in turn influence neighbouring cells, which influence their neighbours, and so on. This biological amplification means that a single exosome treatment can initiate a regenerative cascade that extends across multiple tissue compartments and persists for weeks to months after the initial administration. It is the difference between lighting a match and lighting a fire.
The Core Clinical Distinction: Transient Function vs Sustained Regeneration
The practical difference between peptides and exosomes can be stated simply: peptides serve a specific function at the cellular level, but that function runs out. Exosomes regenerate.
Peptide therapy is best understood as a pharmacological intervention — a targeted, time-limited biological signal that produces a defined physiological response. It is enormously useful for specific clinical goals: accelerating tissue repair after injury, stimulating growth hormone pulsatility, promoting skin collagen synthesis, or supporting neuroprotection. For these applications, peptides are precise, effective, and well-characterised. But peptide therapy does not reprogram the epigenome. It does not reverse the accumulated methylation drift that represents decades of biological aging. When the dosing stops, the biological clock continues ticking at its pre-intervention pace.
Exosome therapy, by contrast, acts at the level of cellular identity. The miRNA cargo delivered by MSC exosomes can regulate the activity of DNA methyltransferases (DNMTs) and TET enzymes — the molecular machinery that writes and erases methylation marks across the genome. This means exosomes have the capacity to directly shift the methylation landscape toward a younger state, not merely to improve the function of aged cells operating within an aged epigenome. The sustained biological effect of a single exosome treatment course — typically 3–6 months of measurable regenerative activity — reflects this deeper mechanism. The cell has not just been instructed to behave differently; it has been given the tools to become different.
The Epigenetic Measurement Question: Which Intervention Moves the Clock?
For the physician who wants to know whether their regenerative protocol is actually reducing biological age — not just improving subjective wellbeing — the answer lies in the DNA methylation data.
| Intervention | Epigenetic Mechanism | First Detectable Clock Signal | Sustained Effect Without Re-dosing |
|---|---|---|---|
| Therapeutic Peptides | Receptor-mediated gene expression changes | 2–3 months (faint) | No — returns toward baseline |
| Exosome Therapy | Direct miRNA-mediated epigenetic reprogramming | 4–6 weeks | Yes — 3–6 months sustained |
| Combined Protocol | Synergistic: peptides for targeted function, exosomes for epigenetic reset | 4–6 weeks (exosome-driven) | Yes — with periodic exosome maintenance |
Peptide therapy can produce a faint epigenetic signal — the downstream gene expression changes triggered by receptor binding do eventually reach the methylation layer — but this signal is modest, slow to develop (typically 2–3 months of consistent dosing), and critically, it reverts toward baseline when dosing stops. The methylation clock does not recognise peptide therapy as a regenerative intervention in the same way it recognises exosome therapy.
Exosome therapy, because it acts directly on the epigenetic machinery through miRNA cargo, produces a measurable shift in validated methylation clocks within 4–6 weeks of treatment. This shift reflects a genuine change in the methylation landscape that can persist for 3–6 months after a single treatment course, as the paracrine cascade continues to propagate regenerative signals through the tissue.
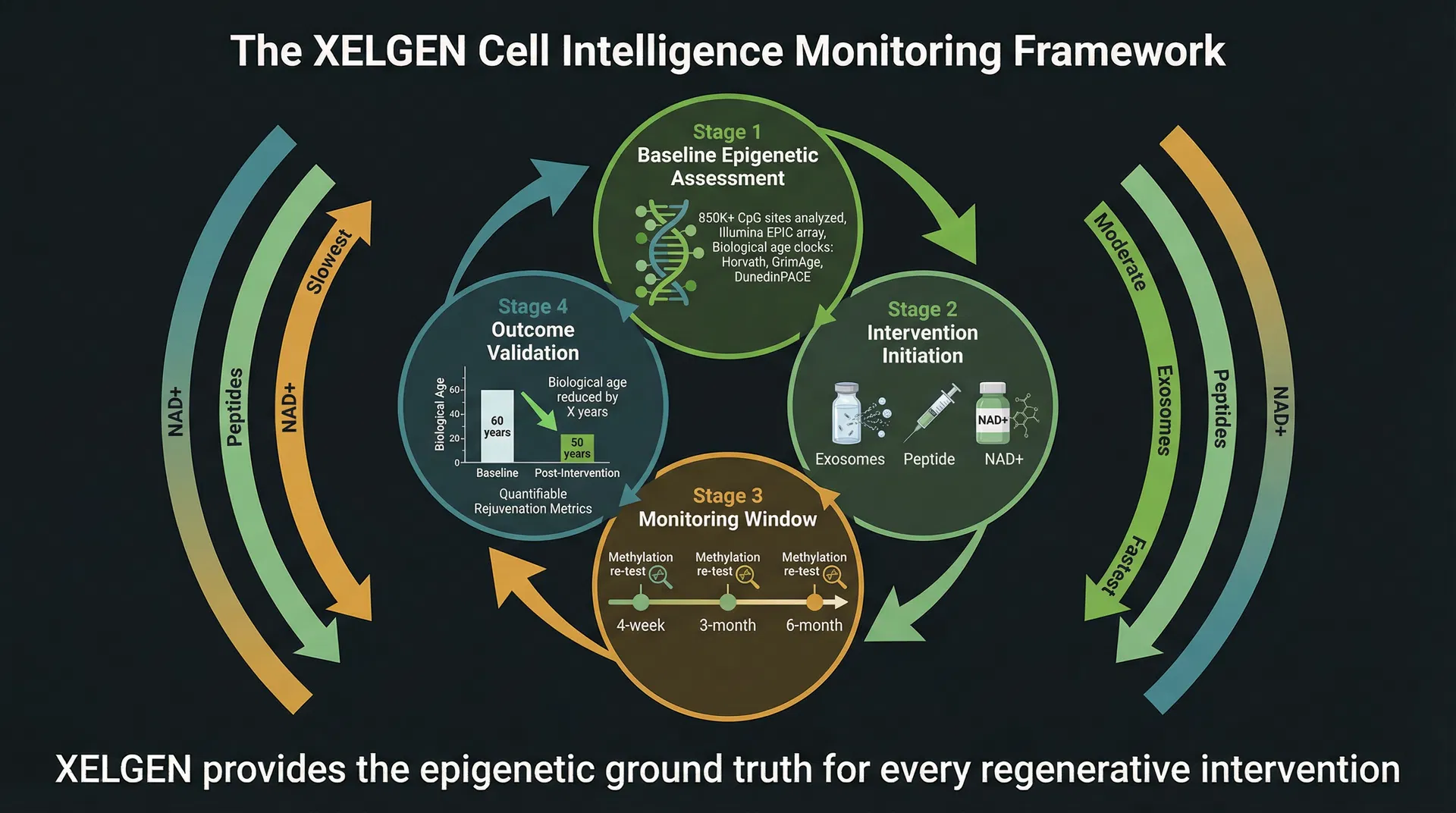
The XELGEN Framework: Measuring What Actually Matters
The XELGEN Cell Intelligence Platform provides genome-wide DNA methylation analysis across more than 850,000 CpG sites using Illumina EPIC array technology. This generates biological age estimates from multiple validated clocks — Horvath's pan-tissue clock, GrimAge, DunedinPACE, and PhenoAge — giving the clinician a comprehensive, multi-dimensional view of where the patient sits on the biological aging spectrum.
For a physician using both peptide therapy and exosome therapy in their practice, XELGEN screening provides the objective data to answer questions that no subjective assessment can address. Is the exosome treatment producing an epigenetic response? A baseline XELGEN screen before treatment, followed by re-testing at 4–6 weeks, will show whether the methylation clock has shifted. Are the peptides contributing to the epigenetic picture? For patients on long-term peptide protocols, XELGEN re-testing at 3–6 months can detect whether the cumulative functional improvements are translating into measurable methylation changes. What is the combined effect of a multi-modal protocol? XELGEN screening provides the only objective measure of the net epigenetic outcome — separating the signal from the noise.
Building the Optimal Regenerative Protocol
The clinical evidence does not support a binary choice between peptides and exosomes. The most effective regenerative protocols use both — but with a clear understanding of what each is contributing.
MSC-derived exosome therapy, administered as a structured treatment course, provides the primary epigenetic intervention. It initiates the regenerative cascade, shifts the methylation clock, and establishes a new biological baseline from which further optimisation can proceed.
Therapeutic peptides are deployed for specific clinical goals alongside the epigenetic reset: BPC-157 for gut integrity and musculoskeletal repair, GHK-Cu for skin and tissue remodelling, growth hormone secretagogues for metabolic optimisation. Precision tools for specific functional deficits.
Baseline and follow-up methylation screening provides the objective data that validates the protocol, guides dose adjustments, and documents outcomes. In a field where patient expectations are high, this measurement layer is the clinical standard that separates evidence-based medicine from unvalidated wellness marketing.
"The future of regenerative medicine belongs to protocols that combine the precision of peptide pharmacology with the regenerative depth of exosome biology, measured against the only ground truth that cannot be faked: what the epigenome actually shows."
XELGEN's genome-wide methylation platform gives your clinic the objective biological age data to validate every intervention — from the first exosome treatment to long-term peptide maintenance protocols.
Request Platform AccessReferences
- Lau, J.L., & Dunn, M.K. (2018). Therapeutic peptides: Historical perspectives, current development trends, and future directions. Bioorganic & Medicinal Chemistry, 26(10), 2700–2707. https://doi.org/10.1016/j.bmc.2017.06.052
- Sikiric, P., et al. (2018). Brain-gut Axis and Pentadecapeptide BPC 157: Theoretical and Practical Implications. Current Neuropharmacology, 16(10), 1523–1548. https://doi.org/10.2174/1570159X15666170619144112
- Goldstein, A.L., Hannappel, E., & Kleinman, H.K. (2005). Thymosin β4: actin-sequestering protein moonlights to repair injured tissues. Trends in Molecular Medicine, 11(9), 421–429. https://doi.org/10.1016/j.molmed.2005.07.004
- Pickart, L., & Margolina, A. (2018). Regenerative and Protective Actions of the GHK-Cu Peptide in the Light of the New Gene Data. International Journal of Molecular Sciences, 19(7), 1987. https://doi.org/10.3390/ijms19071987
- Kalluri, R., & LeBleu, V.S. (2020). The biology, function, and biomedical applications of exosomes. Science, 367(6478), eaau6977. https://doi.org/10.1126/science.aau6977
- Théry, C., et al. (2018). Minimal information for studies of extracellular vesicles 2018 (MISEV2018). Journal of Extracellular Vesicles, 7(1), 1535750. https://doi.org/10.1080/20013078.2018.1535750
- Wiklander, O.P.B., et al. (2019). Advances in therapeutic applications of extracellular vesicles. Science Translational Medicine, 11(492), eaav8521. https://doi.org/10.1126/scitranslmed.aav8521
- Horvath, S., & Raj, K. (2018). DNA methylation-based biomarkers and the epigenetic clock theory of ageing. Nature Reviews Genetics, 19(6), 371–384. https://doi.org/10.1038/s41576-018-0004-3
- Belsky, D.W., et al. (2020). DunedinPACE, a DNA methylation biomarker of the pace of aging. eLife, 11, e73420. https://doi.org/10.7554/eLife.73420
- Lu, A.T., et al. (2019). DNA methylation GrimAge strongly predicts lifespan and healthspan. Aging, 11(2), 303–327. https://doi.org/10.18632/aging.101684
© 2026 XELGEN株式会社 · resources.xelgen.bio
© 2026 XELGEN株式会社 · 〒160-0012 東京都新宿区南元町15-59, JAPAN