The Clinical Evidence Base for Epigenetic Medicine
Peer-reviewed scientific papers, clinical integration guides, and case studies for regenerative medicine clinics. Built on Illumina technology — genome-wide DNA methylation analysis for biological age insights.
Resources for Regenerative Medicine Clinics
Scientific papers, clinical guides, and case studies — curated for stem cell physicians, medical directors, and scientific partners.
Peer-reviewed research on DNA methylation, biological aging, and epigenetic biomarkers.
Step-by-step integration protocols for regenerative medicine and longevity clinics.
Real-world clinical outcomes from stem cell clinics using epigenetic biomarker testing.
Expert commentary, clinical perspectives, and emerging research in longevity medicine.
Core Scientific Publications

XELGEN Methylation Testing: Scientific Overview
A comprehensive overview of genome-wide DNA methylation analysis using the Illumina Infinium EPIC array platform. Covers CpG site selection methodology, biological age clock validation, and clinical interpretation frameworks for regenerative medicine applications.

Epigenetic Biomarkers of Stem Cell Potency
This study identifies epigenetic signatures predictive of mesenchymal stem cell differentiation capacity. Analysis of 850,000 CpG sites reveals methylation patterns correlated with clinical outcomes in regenerative medicine interventions.
Clinical Integration Guide for Regenerative Medicine Clinics
A practical protocol for integrating biological age testing into stem cell clinic workflows. Covers patient selection criteria, pre-treatment baseline assessment, post-treatment monitoring intervals, and reporting standards.
From the XELGEN Scientific Team
Case studies, clinical research, and insights from the XELGEN scientific team
Exosome Therapy for Parkinson's Disease: Epigenomic Results & Case Study
76-year-old female patient. 3-month IV exosome treatment. 269,095 CpG sites analysed. 27 protective genes reactivated. Biological age reversed 2.1 years. Full methylation data, Digital Twin counterfactual model, and clinical protocol recommendations.

🇯🇵 パーキンソン病エクソソーム療法:エピゲノム治療結果と症例研究
76歳女性患者。3ヶ月間のエクソソーム静脈注射療法。269,095 CpGサイト解析。27の保護遺伝子が再活性化。生物学的年齢が2.1年若返り。完全なメチル化データ、デジタルツイン反事実モデル、および臨床プロトコル推奨事項を含む。

NAD+ vs Exosomes: Which Intervention Actually Moves the Needle on Biological Age?
Both NAD+ precursors and exosome therapy are attracting serious attention from regenerative medicine clinicians. But they work through fundamentally different mechanisms, operate on different timescales, and leave very different epigenetic footprints. XELGEN methylation screening is the only objective tool that can tell you which one is actually working — and when.
Exosomes vs Therapeutic Peptides: Understanding the Difference, and Why It Matters for Long-Term Regeneration
Peptides serve a specific function at the cellular level — but that function runs out. Exosomes regenerate. This article examines the science behind each, compares clinical evidence, and explains how XELGEN methylation screening provides the objective measurement layer to determine which intervention is actually moving the biological age needle.

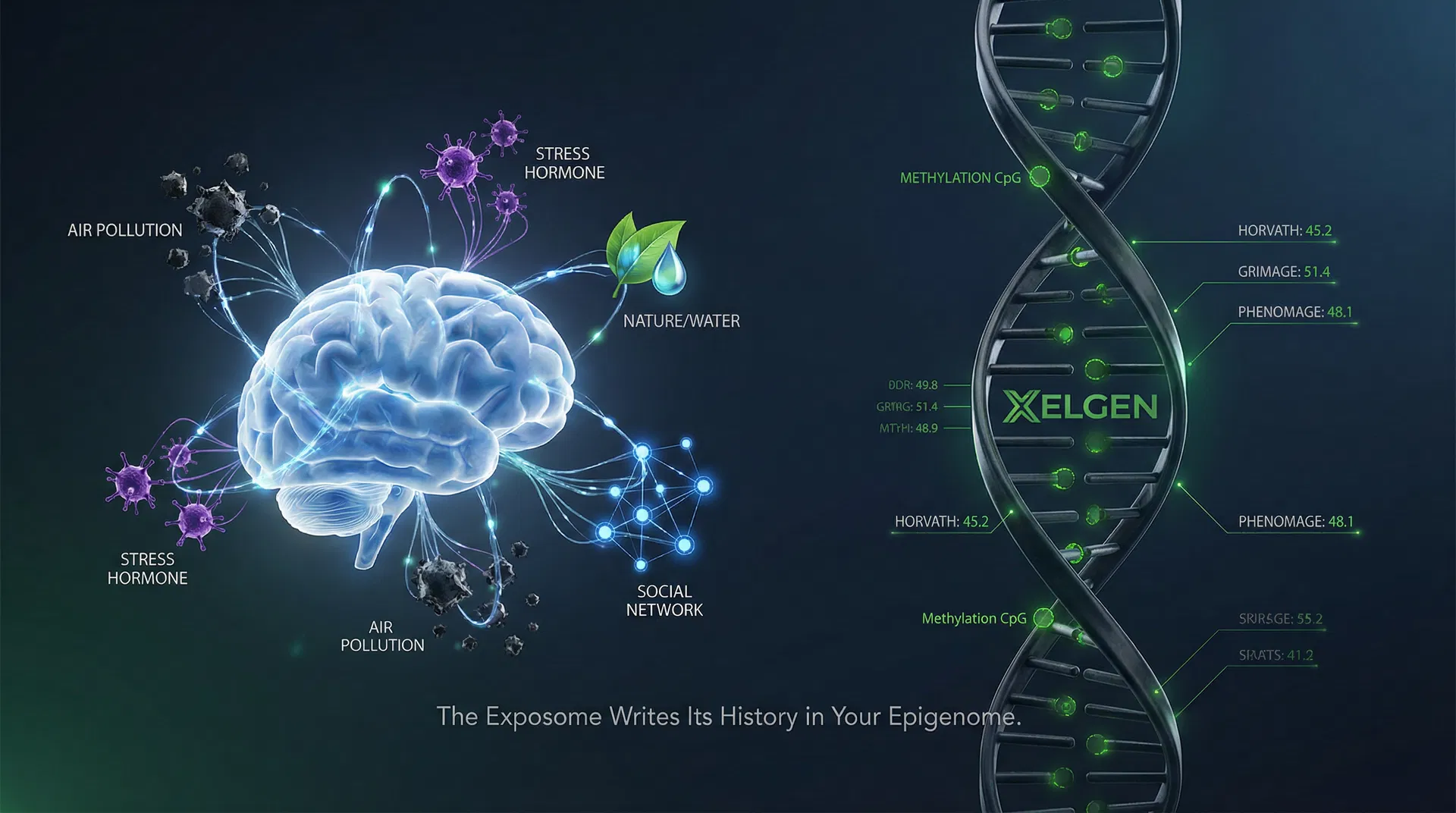
Your Epigenome as a Lifetime Exposure Log: What XELGEN's 850K CpG Screen Reveals
Every environmental exposure leaves a molecular trace in the epigenome — air pollution, diet, stress, and toxins write themselves into DNA methylation marks that persist for decades. XELGEN's 850K CpG platform reads that lifetime record at clinical resolution, translating environmental history into actionable biological age intelligence.

When the Epigenome Loses Its Memory: The Four Pillars of Ageing and What XELGEN Measures
A landmark Nature Reviews framework reframes ageing as systems-level epigenetic fidelity failure across four interacting pillars. XELGEN's 850K CpG platform reads all four failure modes simultaneously — enabling targeted intervention selection and multi-pillar response monitoring.
Why Single Biomarkers Fail: The Case for Multi-Factor Epigenomic Intelligence in Biological Age Assessment
A Nature Medicine study of 18,701 individuals across 34 countries proves that multi-factor exposome interactions explain up to 15× more variance in biological ageing than any single exposure. XELGEN's 850K CpG platform reads all four pathway domains simultaneously — delivering the multi-factor intelligence the science demands.
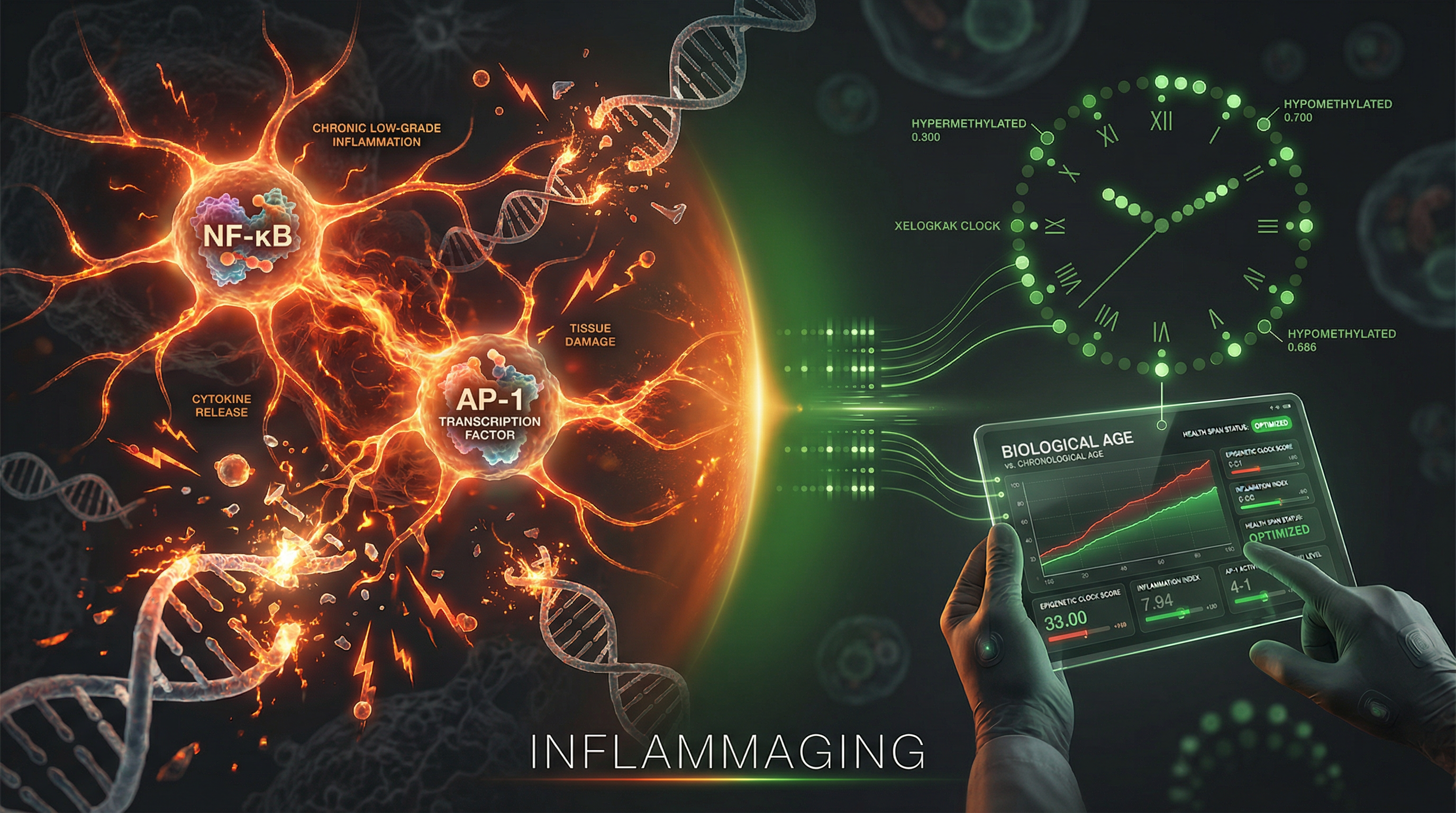
The Silent Fire Within: How XELGEN Detects and Monitors Inflammaging
AP-1 hijacking, Polycomb erosion, and SASP activation are the downstream outputs of all four epigenetic fidelity pillars. XELGEN's Inflammaging Index (XII) — derived from 850K CpG methylation — is the only clinical instrument that measures the fire, tracks its progression, and confirms it is being extinguished under intervention.

Stem Cell Therapy & Epigenetic Response: How to Dose, Time, and Measure
Stem cell therapy is one of the most powerful epigenetic reprogramming tools in regenerative medicine — but without molecular monitoring, clinicians cannot confirm it worked. XELGEN's three-point protocol transforms stem cell therapy into a precision medicine protocol guided by molecular evidence.
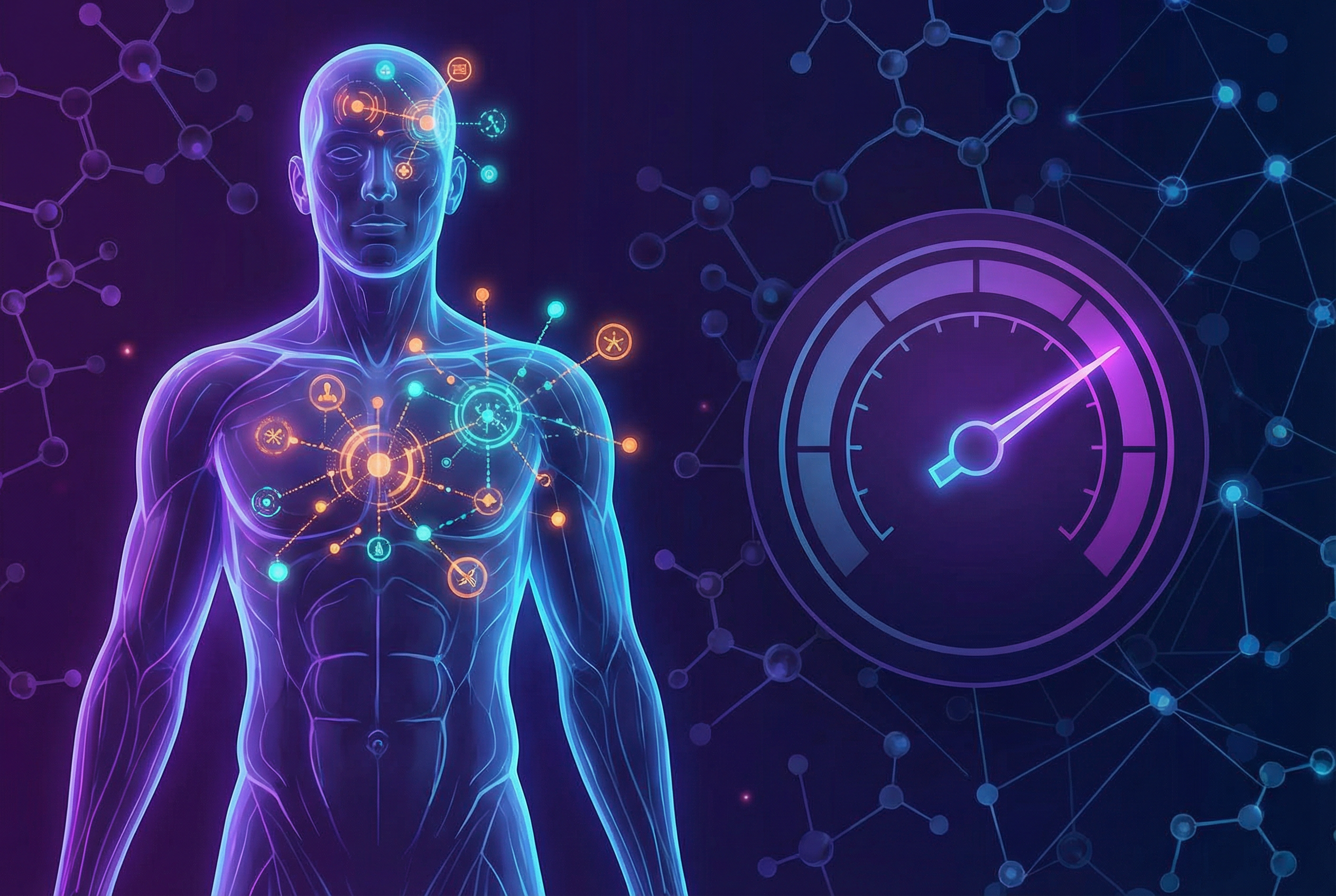
Using Biological Age Biomarkers to Assess Disease Risk in Preventive Medicine
Epigenetic aging acceleration is associated with cardiovascular disease, metabolic syndrome, and all-cause mortality. How biological age biomarkers complement traditional risk assessment tools.

Cellular Senescence: A Key Driver of Aging and Degenerative Disease
Senescent cells accumulate with age and release inflammatory SASP factors that damage surrounding tissues — driving cardiovascular disease, neurodegeneration, and metabolic disorders.
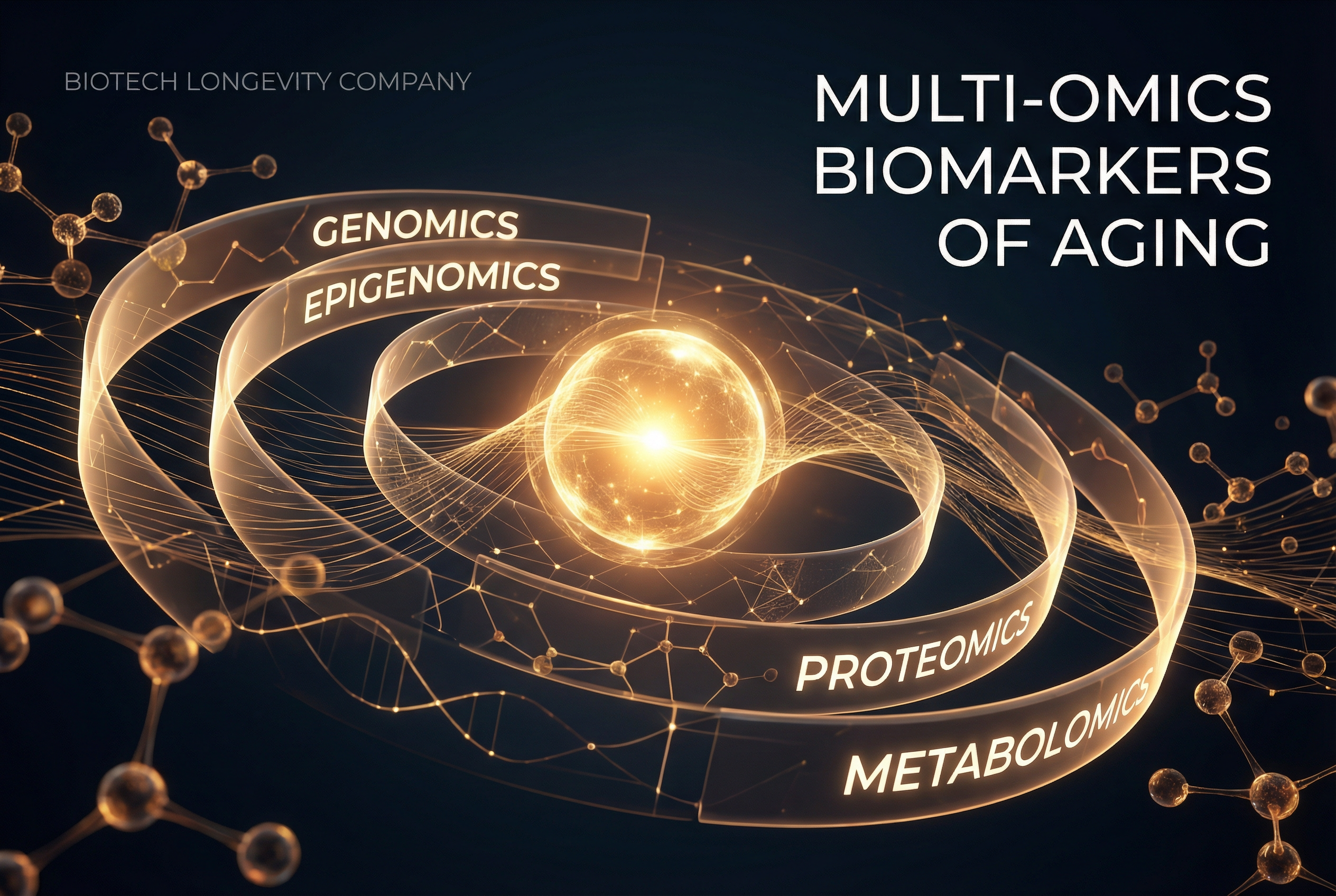
Multi-Omics Biomarkers of Aging: Integrating Genomics, Epigenetics, and Proteomics
Multi-omics approaches combine genomic, epigenetic, proteomic, and metabolomic data to build comprehensive models of biological aging beyond what any single layer can reveal.

Epigenetic Aging Clocks Explained: Horvath, GrimAge, PhenoAge & DunedinPACE
A comprehensive guide to the four major epigenetic aging clocks — how they work, what they predict, and how to choose the right one for your clinical application.

The Role of Epigenetics in Stem Cell Aging
Stem cell function declines with age due to epigenetic drift — systematic changes in DNA methylation that alter self-renewal, differentiation, and regenerative capacity.

How Longevity Clinics Use Biological Age Monitoring
Longitudinal biological age monitoring converts a longevity program from a subjective wellness experience into a measurable, data-driven clinical protocol.

Artificial Intelligence and Biological Age Prediction
Machine learning and multi-omics integration are transforming how we predict biological age — enabling personalized aging trajectories and AI-driven longevity protocols.
Science, Simplified
Accessible deep-dives into epigenetics, longevity science, and clinical practice — written for physicians and their teams.
The Role of Epigenetics in Stem Cell Aging
Stem cell function declines with age due to epigenetic drift — systematic changes in DNA methylation that alter self-renewal, differentiation, and regenerative capacity. A clinical overview for regenerative medicine practitioners.
Cellular Senescence: A Key Driver of Aging and Degenerative Disease
Senescent cells accumulate with age and release inflammatory SASP factors that damage surrounding tissues. Learn how senescence drives cardiovascular disease, neurodegeneration, and metabolic disorders.
Can Biological Aging Be Reversed? What the Science Says
The Fitzgerald trial and TRIIM study suggest epigenetic age can shift in response to targeted interventions. A critical review of the evidence and what it means for clinical longevity practice.
Why Biological Age Can Change Over Time
Biological age is not a fixed score — it fluctuates in response to illness, stress, and recovery. Understanding its dynamic nature is essential for longitudinal monitoring in longevity medicine.
Lectures & Clinical Presentations
On-demand webinars, clinical talks, and research presentations from the XELGEN scientific team.

Understanding DNA Methylation Clocks: A Clinical Primer

Integrating Biological Age Testing into Your Regenerative Medicine Practice

Epigenetic Biomarkers and Stem Cell Potency: What the Data Shows
Curated from Leading Science Publications
Third-party reporting on the science shaping regenerative medicine — with XELGEN context.
 XELGENConference ReportNEW
XELGENConference ReportNEWWhere Biohacking Meets Cell Intelligence — XELGEN at Biohackers World LA 2026
Nearly 2,000 biohackers, longevity researchers, and clinicians gathered in Los Angeles for Biohackers World 2026 — and the conversation around exosome and stem cell therapy has never been more urgent. XELGEN's co-founder and Chief Growth Officer were there, and what they witnessed confirmed everything we are building toward: the market is ready for precision cellular intelligence.
Biohackers World LA was a defining moment for XELGEN in the United States. The passion in that room — from clinicians asking hard questions about exosome potency, to biohackers demanding data behind their therapies — is exactly the energy that drives our work. We are now actively partnering with leading clinics across Los Angeles and San Francisco, bringing the XELGEN Cell Intelligence Platform to the clinicians who are shaping the future of regenerative medicine.
 XELGENConference ReportNEW
XELGENConference ReportNEWXELGEN at the American Academy for Stem Cell Physicians — Miami 2026
XELGEN attended the AASCP Conference in Miami — one of the most important gatherings of stem cell clinicians in the United States. The consensus was clear: stem cell medicine is evolving faster than the clinical frameworks supporting it. The missing piece? Cellular intelligence.
The XELGEN Cell Intelligence Platform has officially landed in the United States. Following AASCP Miami, we are now in active discussions with stem cell clinics across the country to deploy precision epigenetic intelligence in real clinical settings. Next stop: Los Angeles.
 XELGENConference ReportNEW
XELGENConference ReportNEWXELGENがAASCPカンファレンス — マイアミ 2026に参加 🇯🇵
XELGENは、米国で最も重要な幹細胞医療の専門家・研究者が集うAASCPマイアミ大会に参加しました。コンセンサスは明確でした。幹細胞医療は、それを支える臨床フレームワークよりも速く進化しています。欠けているピース、それがセルインテリジェンスです。
XELGENセルインテリジェンス・プラットフォームが米国に正式上陸。AASCPマイアミ参加を経て、全米の幹細胞クリニックと積極的な協議を進めています。次の目的地:ロサンゼルス。
 Harvard Medical SchoolCell IntelligenceNEW
Harvard Medical SchoolCell IntelligenceNEWCombining Biology and AI To Advance Cell Therapy
One of the most enduring goals in regenerative medicine is deceptively simple: replace a person's damaged or dying cells with healthy new ones grown in the laboratory. A Harvard Medical School spinout is now combining deep biological insight with AI to make that goal a clinical reality — accelerating the path from stem cell science to scalable therapy.
This is exactly the convergence XELGEN is built for. Deep cellular knowledge — decoded at the epigenomic level — is the foundation of true cell intelligence. As AI begins to read the language of cells, XELGEN's genome-wide DNA methylation platform provides the biological ground truth that makes those models clinically meaningful. The future of regenerative medicine is not just smarter algorithms; it is algorithms trained on richer cellular data.
 NIEHS / NIHEpigenetics & EnvironmentNEW
NIEHS / NIHEpigenetics & EnvironmentNEWEpigenetics and the Exposome: A New Era of Disease Prevention
NIEHS Director Rick Woychik speaks with Harvard's Andrea Baccarelli about how the epigenome acts as a molecular fingerprint of every environmental exposure we encounter — from air pollution to diet to stress — and how DNA methylation patterns can reveal past exposures and predict disease risk years before symptoms appear.
This is the science that underpins everything XELGEN does. The exposome leaves its marks in DNA methylation — and XELGEN's genome-wide 850K CpG platform reads those marks with clinical precision. Every patient we screen carries an epigenomic record of their lifetime exposures. Our role is to decode that record, quantify biological age acceleration, and give clinicians the intelligence to intervene before disease takes hold.
 Nature Reviews MCBEpigeneticsNEW
Nature Reviews MCBEpigeneticsNEWSystemic Epigenetic Dysregulation as a Driver of Ageing and a Therapeutic Target
Harvard's Vadim Gladyshev proposes a landmark four-pillar framework in Nature Reviews Molecular Cell Biology: ageing is driven by systems-level failure of epigenetic fidelity — nuclear architecture deterioration, epigenetic memory collapse, histone variant drift, and AP-1 transcriptional hijacking — not merely by isolated methylation changes.
This is the theoretical foundation that gives XELGEN's 850K CpG platform its clinical power. When Gladyshev argues that ageing reflects 'collapse of coordinated chromatin regulation,' he is describing exactly what XELGEN measures at genome-wide resolution. Our methylation screen does not just report a biological age number — it reads the fidelity of the entire epigenetic control system across 850,000 CpG sites, giving clinicians a direct window into which of these four failure modes is already active in their patient.
 Nature MedicineBrain Aging & ExposomeNEW
Nature MedicineBrain Aging & ExposomeNEWThe Exposome of Brain Aging Across 34 Countries
The largest global study of its kind — 18,701 individuals across 34 countries — shows that 73 environmental factors (air pollution, climate variability, green space, water quality, socioeconomic inequality, democratic stability) explain up to 15× more variance in brain aging than any single exposure alone. Crucially, the effects are syndemic and nonlinear: interactions across domains amplify biological impact far beyond what any single-factor model can detect.
This landmark finding is the scientific case for what XELGEN does. Single biomarkers, single exposures, single interventions — none of them capture the true biology of aging. XELGEN's 850K CpG methylation platform reads the genome-wide fingerprint of every accumulated exposure simultaneously, mapping how air quality, stress, diet, inflammation, and social environment have interacted to accelerate or protect biological age. When Legaz et al. show that multi-factor interaction explains 15× more variance than any single factor, they are describing exactly why XELGEN's multi-dimensional epigenomic screen is the clinical tool that precision medicine has been waiting for.
 TIMELongevity Science
TIMELongevity ScienceAging Is Medicine's Biggest Blind Spot
ARPA-H's PROSPR program is launching the first Phase 3 clinical trial targeting aging itself — not a single disease — using FDA-approved drugs and a validated biomarker score designed to predict 20-year health outcomes. A $38 trillion opportunity is finally getting the clinical infrastructure it deserves.
ARPA-H's mandate to build validated aging biomarker scores and accessible home testing kits is a direct validation of XELGEN's clinical thesis. Our genome-wide DNA methylation platform already delivers the multi-system biological age intelligence that PROSPR is working to standardise — positioning XELGEN as the epigenetic layer that longevity medicine needs right now, not in 2031.
 Clinical EpigeneticsEpigenetic Research
Clinical EpigeneticsEpigenetic ResearchEpigenetic Patterns, Accelerated Biological Aging & Epigenetic Drift Detected 6 Months Following COVID-19 Infection
A genome-wide DNA methylation study of 96 post-COVID-19 patients found significant epigenetic age acceleration via Horvath's clock, 42 dysregulated CpG sites, and a substantial increase in stochastic epigenetic mutations — pointing to lasting biological damage across immune, vascular, and metabolic pathways, regardless of initial infection severity.
Biological ageing is not solely a function of time — it is actively shaped by conditions like COVID-19, which can accelerate epigenetic clocks and trigger lasting drift across immune, vascular, and metabolic pathways. XELGEN's genome-wide DNA methylation platform provides the most comprehensive and personalised epigenetic screening available, identifying current biological age acceleration and early signals of future risk before they manifest as disease.
 TIMELongevity Science
TIMELongevity ScienceThe Race to Measure Aging — And Why It Matters
Scientists are racing to develop reliable biological age tests — and the results could reshape how we think about health, disease, and longevity. DNA methylation clocks are emerging as the most validated approach.
This TIME feature validates the clinical demand for epigenetic age measurement. XELGEN's genome-wide DNA methylation platform — built on Illumina EPIC arrays — delivers exactly the validated, physician-grade biological age insights the field is calling for.
 BioPharma DiveCell Therapy
BioPharma DiveCell TherapyWith FDA Go-Ahead, a China Biotech Notches a First in Cell Therapy Testing
The FDA has cleared Shanghai-based Unixell to begin human studies of an allogeneic, stem cell-derived therapy targeting focal epilepsy — marking a new frontier for cell therapy indications globally.
Stem cell therapies for diverse disease indications are set to explode. What the field critically needs is XELGEN's Cell Intelligence platform to ensure cellular changes are continuously monitored, validated, and clinically documented throughout treatment.
 Endpoints NewsGene Therapy
Endpoints NewsGene TherapyFDA Greenlights Life Biosciences' Gene Therapy Study to Rewind the Age of Cells
In a major test for the longevity field, the FDA has cleared Life Biosciences to study a gene therapy designed to achieve a 'near total reset' of cellular age — co-founded by Harvard's David Sinclair.
Advancements in cell and gene therapy are pushing the boundaries of what's biologically possible. Yet there is a critical gap: the lack of standardized monitoring systems. XELGEN's Cell Intelligence platform provides exactly the epigenetic tracking infrastructure needed to document and validate these cellular age changes in real time.
 Fierce BiotechBiotech Investment
Fierce BiotechBiotech Investment3 New Venture Funds Collect Nearly $1B for Biotech in US & EU
A16z, BioNTech/Penn, and Servier Ventures have collectively raised nearly $1 billion in new biotech-focused funds, signaling strong investor conviction in next-generation life sciences platforms.
The surge in biotech venture capital reflects growing investor confidence in precision medicine platforms. XELGEN is positioned at the intersection of epigenomics, regenerative medicine, and clinical diagnostics — exactly the convergence point that top-tier funds are actively seeking to back.
 STAT NewsRegenerative Medicine
STAT NewsRegenerative MedicineNY Giants Team Doctor: Clinics Offering 'Miracle Injections' Are Selling False Hope
The head team physician for the New York Giants warns that regenerative medicine marketing has outpaced the science — and that unregulated stem cell clinics are putting patients at serious risk.
This is precisely why objective, validated cell therapy screening is essential. XELGEN's Cell Intelligence platform provides the epigenetic monitoring infrastructure that ensures regenerative therapies are not only administered — but rigorously tracked, validated, and proven effective for each individual patient.
Conference Reports
First-hand dispatches from the events shaping regenerative medicine.

Where Biohacking Meets Cell Intelligence
Nearly 2,000 biohackers, longevity researchers, and clinicians gathered in LA — and the conversation around exosome and stem cell therapy has never been more urgent.

Where Stem Cell Science Meets Clinical Practice
XELGEN attended the 2nd International Conference on Stem Cells — one of the most important gatherings of stem cell clinicians in the United States.

幹細胞科学と臨床実践の交差点
XELGENはマイアミで開催された第2回国際幹細胞学会に参加しました。再生医療の最前線で活躍する臨床医・研究者が集結したこの会議から。
日本語医師向けリソース
再生医療の最前線——XELGENオリジナル科学論文、症例研究、臨床ガイド

NAD+とエクソソーム:生物学的年齢に本当に変化をもたらすのはどちらか?
NAD+前駆体とエクソソーム療法は根本的に異なるメカニズムで作用し、エピゲノムに異なる痕跡を残します。XELGENメチル化スクリーニングだけが、どちらが実際に効果をもたらしているかを客観的に判断できます。

エクソソームと治療用ペプチド:違いを理解し、長期的な再生にとってなぜ重要なのか
ペプチドは細胞レベルで特定の機能を果たしますが、最終的には尽きます。エクソソームは再生します。XELGENメチル化スクリーニングが、どちらの介入が生物学的年齢の針を動かしているかを測定します。

エピゲノムは生涯の曝露記録:XELGENの850K CpGスクリーニングが明らかにするもの
大気汚染、食事、ストレス、有害物質——あらゆる環境曝露はエピゲノムに分子的な痕跡を残します。XELGENの850K CpGプラットフォームはその生涯の記録を臨床的解像度で読み取り、環境的歴史を実用的な生物学的年齢インテリジェンスへと変換します。

エピゲノムが記憶を失うとき:老化の四つの柱とXELGENが測定するもの
Nature誌の画期的なフレームワークが老化をシステムレベルのエピジェネティック忠実性崩壊として再定義。核構造の劣化、エピジェネティック記憶の崩壊、ヒストンバリアントのドリフト、AP-1転写ハイジャック——XELGENの850K CpGプラットフォームはこれらすべての崩壊モードを同時に読み取ります。

単一バイオマーカーが失敗する理由:生物学的年齢評価における多因子エピゲノム・インテリジェンスの必要性
Nature Medicine誌の34カ国、18,701名の研究が証明:多因子エクスポソーム相互作用は生物学的老化における分散を単一曝露の最大15倍説明する。XELGENの850K CpGプラットフォームは4つの経路ドメインを同時に読み取り、科学が求める多因子インテリジェンスを提供します。

内なる静かな炎:XELGENが炎症性老化を検出・監視する方法
AP-1転写ハイジャック、ポリコーム侵食、SASP活性化——これらはエピジェネティック忠実性崩壊の4つの柱すべての下流アウトプットです。XELGENの炎症性老化指数(XII)は850K CpGメチル化から導出され、炎症を測定し、その進行を追跡し、介入によって消火されていることを確認する唯一の臨床ツールです。

幹細胞療法とエピジェネティック反応:投与量・タイミング・測定の科学
幹細胞療法は再生医療で最も強力なエピジェネティック再プログラミングツールの一つです——しかし分子モニタリングなしでは、臨床医は効果を確認できません。XELGENの3点プロトコル(ベースライン・6〜8週・5〜6ヶ月)が幹細胞療法を精密医療プロトコルへと変換します。

エピジェネティクスとエクスポソーム:疾病予防の新時代
NIEHSのリック・ウォイチク所長とハーバード大学のアンドレア・バカレッリ教授が、エピゲノムが環境曝露の「分子指紋」として機能し、DNAメチル化パターンが症状が現れる何年も前に疾患リスクを予測できることを解説。XELGENの850K CpGプラットフォームは、この科学を臨床精度で実現します。

🇯🇵 パーキンソン病エクソソーム療法:エピゲノム治療結果と症例研究
76歳女性患者。3ヶ月間のエクソソーム静脈注射療法。269,095 CpGサイト解析。27の保護遺伝子が再活性化。生物学的年齢が2.1年若返り。完全なメチル化データと臨床プロトコル推奨事項を含む。
XELGENは日本の再生医療クリニックと提携し、エピゲノム全体のDNAメチル化解析を通じて生物学的年齢の客観的測定を提供しています。 詳細は xelgen.bio をご覧ください。
Get curated science news + XELGEN context
New articles from the XELGEN Science Team, curated press from leading journals, and clinical insights — delivered to your inbox. For physicians, researchers, and longevity clinicians.
Bring Epigenetic Biomarkers to Your Clinic
Join over 200 regenerative medicine clinics using XELGEN's genome-wide DNA methylation analysis to deliver biological age insights to their patients.
For licensed healthcare providers and medical institutions only · U.S. access