
All Articles & Insights
Peer-reviewed science, clinical education, and longevity insights authored by XELGEN Science Team — Original Article.
What Is Biological Age and Why It Matters for Healthspan
Biological age reflects the cumulative effects of genetics, lifestyle, and environment on the body's systems — and it can differ dramatically from chronological age.

Biological Age vs Chronological Age: Why Aging Happens at Different Rates
Two people born the same year can have biological ages 20 years apart. The molecular mechanisms behind differential aging rates and what this means for clinical practice.

How DNA Methylation Measures Biological Aging
DNA methylation patterns change systematically across the genome as humans age. How epigenetic clocks use these patterns to estimate biological age with clinical-grade accuracy.

Epigenetic Aging Clocks Explained: Horvath, GrimAge, PhenoAge & DunedinPACE
A comprehensive guide to the four major epigenetic aging clocks — how they work, what they predict, and how to choose the right one for your clinical application.

Why DNA Methylation Is the Leading Aging Biomarker
Among all molecular biomarkers of aging, DNA methylation stands out for its precision, reproducibility, and clinical applicability. A scientific comparison of the leading aging biomarker classes.

Epigenetics and the Exposome: A New Era of Disease Prevention
NIEHS Director Rick Woychik and Harvard's Andrea Baccarelli discuss how the epigenome acts as a molecular fingerprint of every environmental exposure — from air pollution to diet — and how DNA methylation patterns can predict disease risk years before symptoms appear.

Your Epigenome as a Lifetime Exposure Log: What XELGEN's 850K CpG Screen Reveals
Every environmental exposure leaves a molecular trace in the epigenome — air pollution, diet, stress, and toxins write themselves into DNA methylation marks that persist for decades. XELGEN's 850K CpG platform reads that lifetime record at clinical resolution.

The Role of Epigenetics in Stem Cell Aging
Stem cell function declines with age due to epigenetic drift — systematic changes in DNA methylation that alter self-renewal, differentiation, and regenerative capacity.

Cellular Senescence: A Key Driver of Aging and Degenerative Disease
Senescent cells accumulate with age and release inflammatory SASP factors that damage surrounding tissues — driving cardiovascular disease, neurodegeneration, and metabolic disorders.

How Lifestyle and Stress Influence Epigenetic Aging
Diet, sleep, stress, and environmental exposures leave measurable epigenetic signatures. How lifestyle factors accelerate or decelerate biological aging and what this means for longevity medicine.

Can Biological Aging Be Reversed? What the Science Says
The Fitzgerald trial and TRIIM study suggest epigenetic age can shift in response to targeted interventions. A critical review of the evidence and clinical implications.

Why Biological Age Can Change Over Time
Biological age is not a fixed score — it fluctuates in response to illness, stress, and recovery. Understanding its dynamic nature is essential for longitudinal monitoring.

NAD+ vs Exosomes: Which Intervention Actually Moves the Needle on Biological Age?
Both NAD+ precursors and exosome therapy attract serious clinical attention — but they work through fundamentally different mechanisms, operate on different timescales, and leave very different epigenetic footprints. XELGEN methylation screening is the only objective tool that can tell you which one is actually working.
Exosomes vs Therapeutic Peptides: Understanding the Difference, and Why It Matters for Long-Term Regeneration
Peptides serve a specific function at the cellular level — but that function runs out. Exosomes regenerate. This article examines the science behind each, compares clinical evidence, and explains how XELGEN methylation screening provides the objective measurement layer.

When the Epigenome Loses Its Memory: The Four Pillars of Ageing and What XELGEN Measures
A landmark Nature Reviews framework reframes ageing as systems-level epigenetic fidelity failure across four interacting pillars. XELGEN's 850K CpG platform reads all four failure modes simultaneously — enabling targeted intervention selection and multi-pillar response monitoring.

Artificial Intelligence and Biological Age Prediction
Machine learning and multi-omics integration are transforming how we predict biological age — enabling personalized aging trajectories and AI-driven longevity protocols.
Multi-Omics Biomarkers of Aging: Integrating Genomics, Epigenetics, and Proteomics
Multi-omics approaches combine genomic, epigenetic, proteomic, and metabolomic data to build comprehensive models of biological aging beyond what any single layer can reveal.

Japan's Regenerative Medicine Regulatory Framework: A Model for Clinical Innovation
Japan's conditional approval pathway and certified review committee system represent a globally influential model for balancing clinical innovation with patient safety.

Why Standardized Biomarker Data Matters for Longevity Research
Standardized biomarker datasets are essential for reproducible research, cross-study validation, and regulatory acceptance — the foundation of evidence-based longevity medicine.

The Future of Longevity Medicine: Biomarkers, AI, and Precision Aging Science
Longevity medicine is evolving toward individualized, biomarker-driven protocols that measure biological aging at the molecular level and evaluate interventions with scientific rigor.
Using Biological Age Biomarkers to Assess Disease Risk in Preventive Medicine
Epigenetic aging acceleration is associated with cardiovascular disease, metabolic syndrome, and all-cause mortality. How biological age biomarkers complement traditional risk assessment tools.
Biological Age and Cardiovascular Health: Epigenetic Biomarkers of Vascular Aging
Vascular aging is not simply a function of time — it is a function of biological aging rate. How epigenetic biomarkers capture cardiovascular aging processes at the molecular level.

How Longevity Clinics Use Biological Age Monitoring
Longitudinal biological age monitoring converts a longevity program from a subjective wellness experience into a measurable, data-driven clinical protocol.

Longevity Biomarkers: The Future of Preventive Medicine
Precision longevity medicine is emerging as a new clinical paradigm — where biomarker-driven insights support personalized health optimization before disease develops.
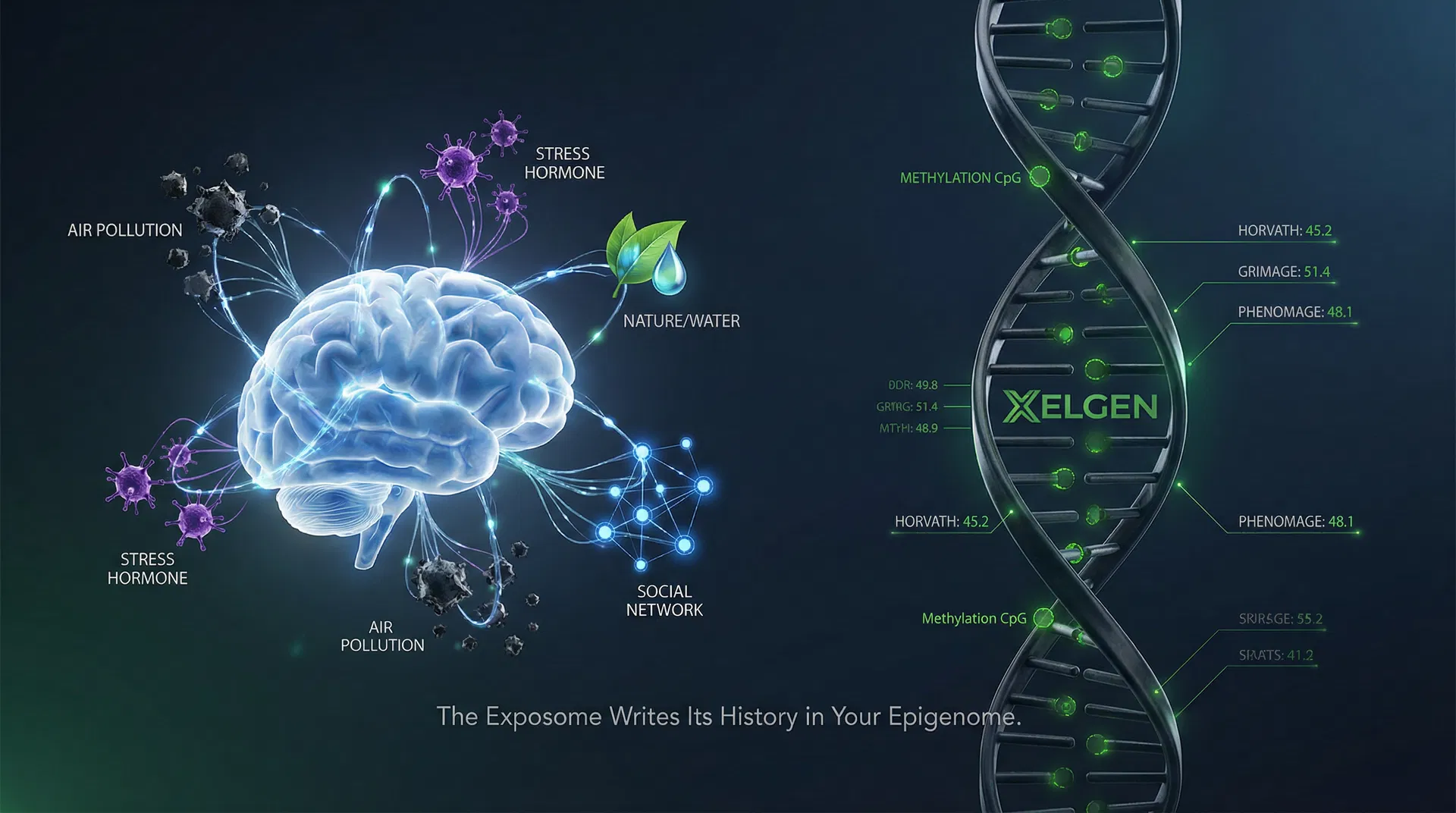
The Exposome of Brain Aging Across 34 Countries
A landmark Nature Medicine study of 18,701 individuals across 34 countries demonstrates that multi-factor environmental exposures explain up to 15× more variance in brain biological age than any single exposure — confirming that ageing is syndemic, nonlinear, and inherently multi-factorial.

Why Single Biomarkers Fail: The Case for Multi-Factor Epigenomic Intelligence in Biological Age Assessment
A Nature Medicine study of 18,701 individuals across 34 countries proves that multi-factor exposome interactions explain up to 15× more variance in biological ageing than any single exposure. XELGEN's 850K CpG platform reads all four pathway domains simultaneously — delivering the multi-factor intelligence the science demands.
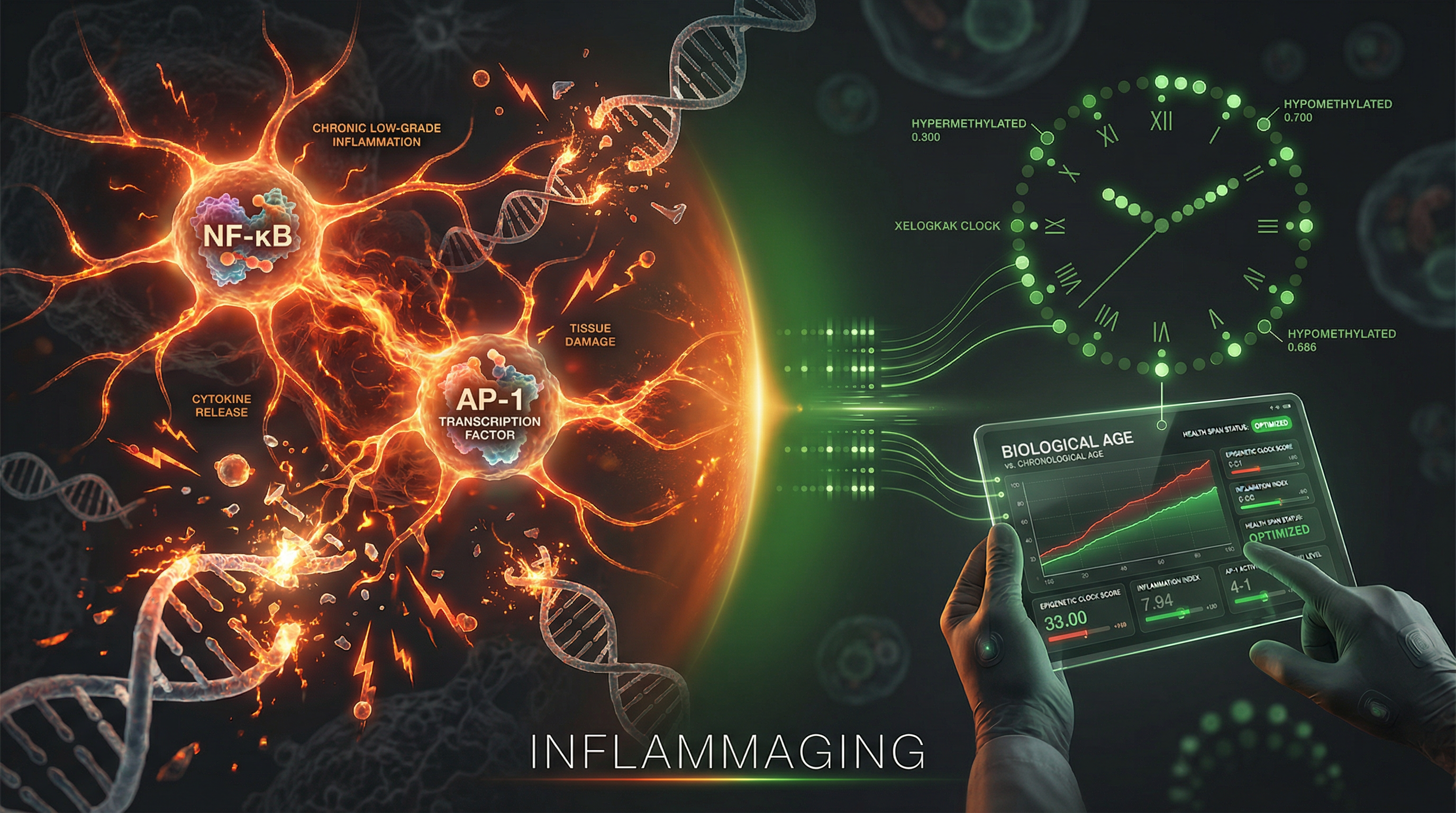
The Silent Fire Within: How XELGEN Detects and Monitors Inflammaging
AP-1 hijacking, Polycomb erosion, and SASP activation are the downstream outputs of all four epigenetic fidelity pillars. XELGEN's Inflammaging Index (XII) — derived from 850K CpG methylation — is the only clinical instrument that measures the fire, tracks its progression, and confirms it is being extinguished under intervention.

内なる静かな炎:XELGENが炎症性老化を検出・監視する方法
AP-1転写ハイジャック、ポリコーム侵食、SASP活性化——これらはエピジェネティック忠実性崩壊の4つの柱すべての下流アウトプットです。XELGENの炎症性老化指数(XII)は850K CpGメチル化から導出され、炎症を測定し、その進行を追跡し、介入によって消火されていることを確認する唯一の臨床ツールです。

Stem Cell Therapy & Epigenetic Response: How to Dose, Time, and Measure
Stem cell therapy is one of the most powerful epigenetic reprogramming tools in regenerative medicine — but without molecular monitoring, clinicians cannot confirm it worked. XELGEN's three-point protocol transforms stem cell therapy into a precision medicine protocol guided by molecular evidence.

幹細胞療法とエピジェネティック反応:投与量・タイミング・測定の科学
幹細胞療法は再生医療で最も強力なエピジェネティック再プログラミングツールの一つです——しかし分子モニタリングなしでは、臨床医は効果を確認できません。XELGENの3点プロトコルが幹細胞療法を精密医療プロトコルへと変換します。

Where Stem Cell Science Meets Clinical Practice: AASCP Miami 2026
XELGEN attended the 2nd International Conference on Stem Cells in Miami — a gathering of leading regenerative medicine clinicians, researchers, and innovators. Here is what we observed, discussed, and brought home.

幹細胞科学と臨床実践の交差点:AASCP マイアミ 2026
XELGENはマイアミで開催された第2回国際幹細胞学会(AASCP)に参加しました。再生医療の最前線で活躍する臨床医・研究者・イノベーターが集結したこの会議から、私たちが持ち帰ったものをお伝えします。

Where Biohacking Meets Cell Intelligence
XELGEN's co-founder and Chief Growth Officer attended Biohackers World 2026 in Los Angeles — nearly 2,000 biohackers, clinicians, and founders gathered to push the frontier of cellular health. Here is what we saw, and why it confirms everything we are building.

バイオハッキングとセルインテリジェンスの交差点
XELGENの共同創業者とChief Growth OfficerがBiohackers World 2026 ロサンゼルスに参加。約2,000名のバイオハッカー、臨床医、起業家が集結した現場から、私たちが持ち帰ったものをお伝えします。